There are 118 "squares" on the periodic table.
The periodic table contains information about the different elements that make up all the solids, liquids, and gases in the known universe.
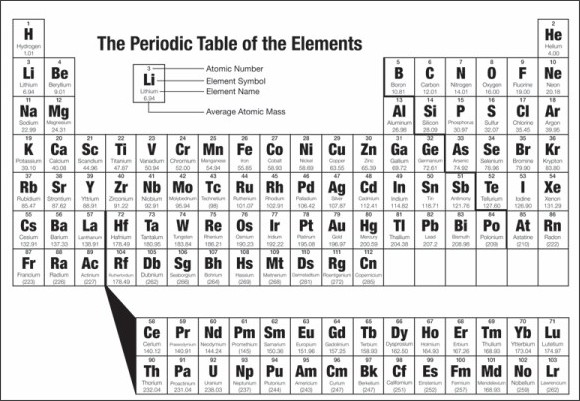
The Periodic Table via kwout
Elements are arranged in order of increasing atomic number. Order generally coincides with increasing atomic mass.
The different rows of elements are called periods. The period number of an element signifies the highest energy level an electron in that element occupies (in the unexcited state), according to the Los Alamos National Laboratory. The number of electrons in a period increases as one moves down the periodic table; therefore, as the energy level of the atom increases, the number of energy sub-levels per energy level increases.
Learn more about the Periodic Table at Livescience.com.
