If a capped syringe is plunged into cold water, the syringe piston will slide inside the syringe. And if a capped syringe is heated, will the syringe plunger will move out of the syringe.
via kwout
Decreasing the temperature inside the syringe by plunging it into the cold water will decrease the molecular activity of the air inside, and therefore it will take up less space.
There are three fundamental gas laws that describe the relationship between pressure, temperature, volume, and amount of gas.
Boyle's Law tells us that the volume of gas increases as the pressure decreases. Charles's Law tells us that the volume of gas increases as the temperature increases. Finally, Avogadro's Law tell us that the volume of gas increases as the amount of gas increases. The ideal gas law is the combination of the three simple gas laws.
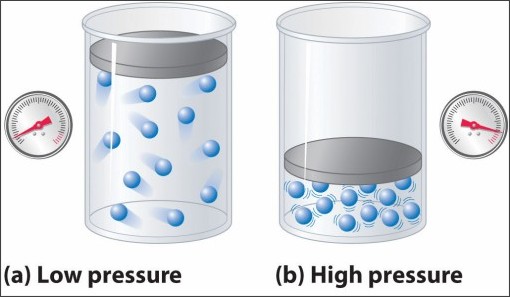
When the volume decrease, the pressure increases. Conversely, when the volume increases, the pressure decreases. This is what is known as an inverse relationship and is demonstrated in Boyle’s Law.
For further reading, see www.esciencelabs.com - Using the Ideal Gas Law.
